
REHOVOT, ISRAEL—August 2, 2023—Scientific excellence requires diversity – research conducted by people from different backgrounds and with varied worldviews. The need for diversity extends to scientific experiments themselves, but the vast majority of studies in the life sciences are conducted using male mice only, which could not only bias the findings, but impede our ability to extrapolate from them to humans. A new study by researchers from the Weizmann Institute of Science addresses this challenge, revealing in unprecedented detail how the brains of male and female mice respond differently to stress.
In the study, published in Cell Reports, researchers from Prof. Alon Chen’s joint laboratory at the Weizmann Institute and the Max Planck Institute of Psychiatry in Munich discovered that a subcategory of brain cells responds to stress differently in males than in females. The findings could lead to a better understanding of health conditions affected by chronic stress, such as anxiety, depression, and even obesity and diabetes; and they could pave the way toward personalized therapies for these disorders.
Mental and physical disorders caused by chronic stress are constantly on the rise, putting a significant strain on society. They affect everyone, but not necessarily in the same ways. Although plenty of evidence suggests that men and women deal differently with stress, the causes of these variances are not yet fully understood, and personalized treatments for men and women are still beyond the reach of medicine. But researchers from Chen’s laboratory, which specializes in studying the response to stress, hypothesized that innovative research methods could help to change the picture. Previous studies in other labs have uncovered certain sex distinctions in the response to stress, but those findings used research methods that could mask, or even entirely erase, the roles played by relatively rare cells. In contract, Chen’s laboratory uses advanced methods that allow scientists to analyze brain activity at an unprecedented resolution – on the level of the individual cell – and could therefore shed new light on the differences between the sexes.
“We turned the most sensitive research lens possible onto the area of the brain that acts as a central hub of the stress response in mammals, the paraventricular nucleus (PVN) of the hypothalamus,” says Dr. Elena Brivio, who led the study. “By sequencing the RNA molecules in that part of the brain on the level of the individual cell, we were able to map the stress response in male and female mice along three main axes: how each cell type in that part of the brain responds to stress, how each cell type previously exposed to chronic stress responds to a new stress experience and how these responses differ between males and females.”
The researchers mapped out gene expression in more than 35,000 individual cells, generating a massive amount of data, providing a picture of stress response unprecedented in its scope, and in highlighting the differences between how males and females perceive and process stress. As part of the study, and in keeping with the principles of open-access science, the researchers decided to make the entire detailed mapping publicly available on a dedicated interactive website, which went live at the same time the study was published, providing other researchers with convenient, user-friendly access to the data. “The website will, for example, allow researchers who are focusing on a specific gene to see how that gene’s expression changes in a certain cell type in response to stress, in males as well as females,” Brivio explains.
The comprehensive mapping has already allowed the researchers to identify a long list of differences in gene expression – between males and females, and between chronic and acute stress. The data showed, inter alia, that certain brain cells respond differently to stress in males and females: Some cells are more susceptible to stress in females and some to stress in males. The most significant difference was found in a type of brain cell called the oligodendrocyte – a subtype of glial cell that provides support to nerve cells and plays an important role in regulating brain activity. In males, exposure to stress conditions, especially chronic stress, changed not only the gene expression in these cells and their interactions with surrounding nerve cells but also their very structure. In females, however, no significant change was observed in these cells, and they were not susceptible to stress exposure.
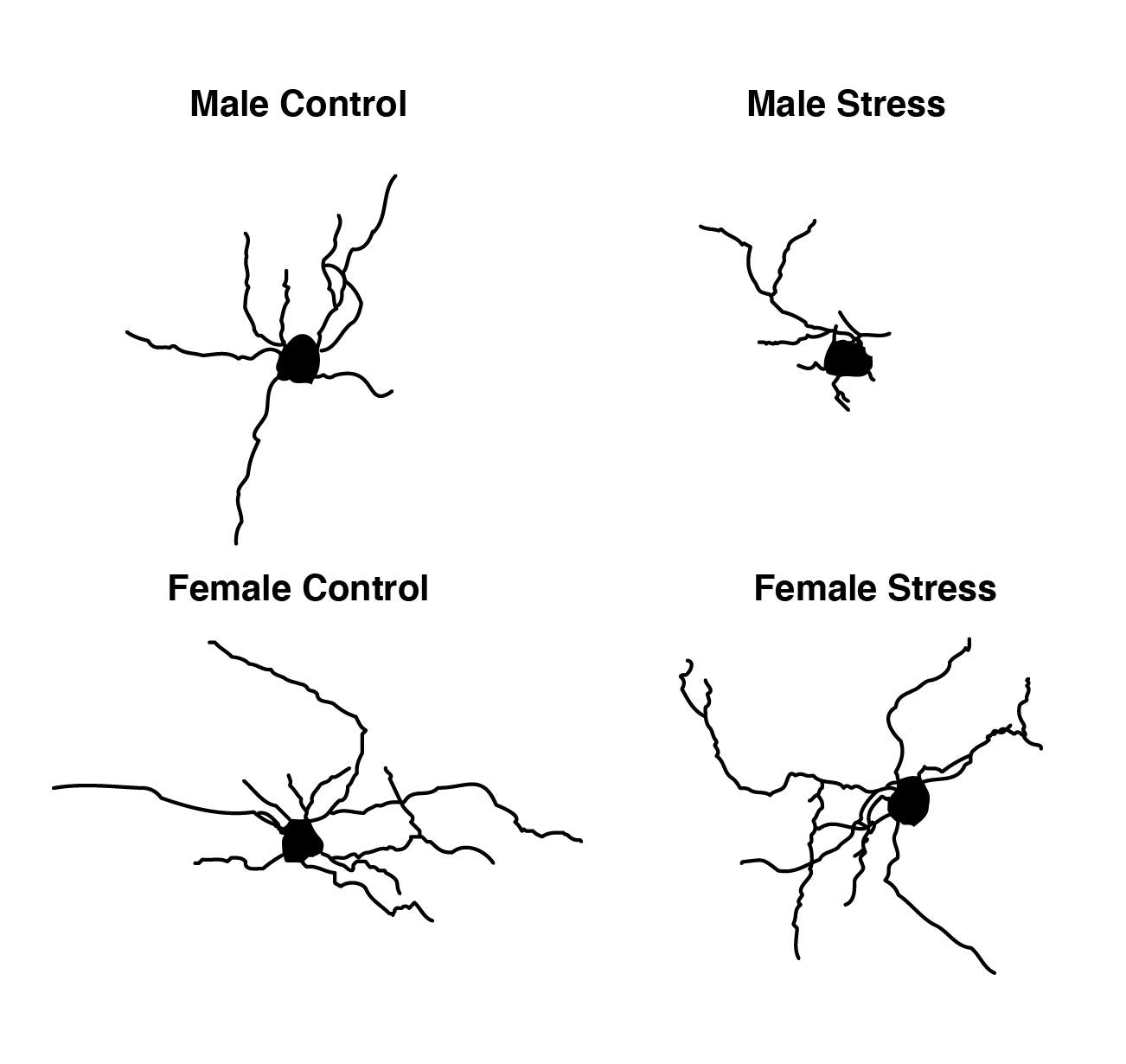
“Neurons attract most of the scientific attention, but they only make up approximately a third of all cells in the brain. The method we implemented allows us to see a much richer and fuller picture, including all the cell types and their interactions in the part of the brain under study,” says Dr. Juan Pablo Lopez, a former postdoctoral fellow in Chen’s group and now the head of a research group at the Department of Neuroscience of the Karolinska Institute in Sweden.
Basic diversity
Until the 1980s, clinical trials of new drugs were conducted on men alone. The accepted view was that including women was unnecessary, and that it would only complicate the research, bringing into play new variables such as menstruation and hormonal changes. For the same reasons, until very recently, preclinical studies avoided using female animals. But it’s now known that the variability among male animals, on a molecular and behavioral level, is usually greater than among females, so there is no reason to suppose that females would complicate the experiments any more than males. Nonetheless, in basic research it’s still common to conduct experiments only on males.
“Our findings show that, when it comes to stress-related health conditions, from depression to diabetes, it’s very important to take the sex variable into account, since it has a significant impact on how different brain cells respond to stress,” Chen explains.
“Even if a study does not specifically focus on the differences between males and females, it’s essential to include female animals in the research, especially in neuroscience and behavioral science, just as it is important to implement the most sensitive research methods, in order to obtain as complete a picture of brain activity as possible,” Brivio adds.

---
Prof. Alon Chen’s research is supported by the Ruhman Family Laboratory for Research in the Neurobiology of Stress; the Licht family; and the Irving B. Harris Fund for New Directions in Brain Research. Prof. Chen is the incumbent of the Vera and John Schwartz Professorial Chair in Neurobiology.
Also participating in the study were Dr. Aron Kos, Stoyo Karamihalev, Andrea Ressle, Rainer Stoffel and Dr. Mathias V. Schmidt of the Max Planck Institute of Psychiatry in Munich; Dr. Alessandro Francesco Ulivi of the Leibniz Institute for Neurobiology in Magdeburg, Germany; Dana Hirsch of Weizmann’s Veterinary Resources Department; and Dr. Gil Stelzer of Weizmann’s Life Sciences Core Facilities Department.
